Homogeneous Palladium Catalyst Suppressing Pd Black Formation in Air Oxidation of Alcohols | Journal of the American Chemical Society

Formation and propagation of well-defined Pd nanoparticles (PdNPs) during C–H bond functionalization of heteroarenes: are nanoparticles a moribund form of Pd or an active catalytic species? - ScienceDirect

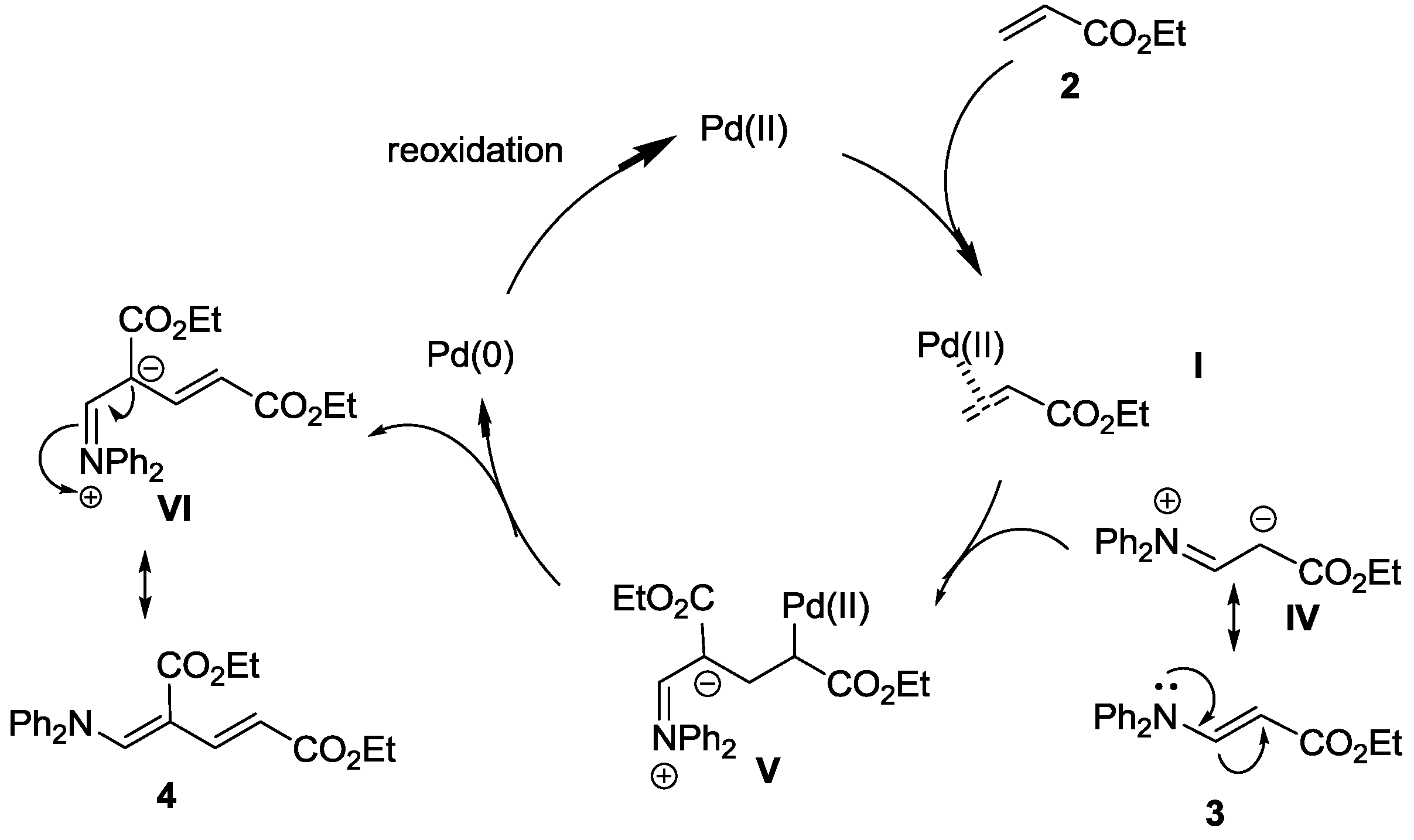
Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics
Catalysts | Free Full-Text | Palladium-Catalyzed Intermolecular Oxidative Amination of Alkenes with Amines, Using Molecular Oxygen as Terminal Oxidant | HTML

Palladium-Catalyzed β-Elimination of Aminoboranes from (Aminomethylsilyl)boranes Leading to the Formation of Silene Dimers | Organometallics

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

Molecules | Free Full-Text | Palladium-Catalyzed Organic Reactions Involving Hypervalent Iodine Reagents | HTML

Catalyst Activation, Deactivation, and Degradation in Palladium‐Mediated Negishi Cross‐Coupling Reactions - Böck - 2015 - Chemistry – A European Journal - Wiley Online Library
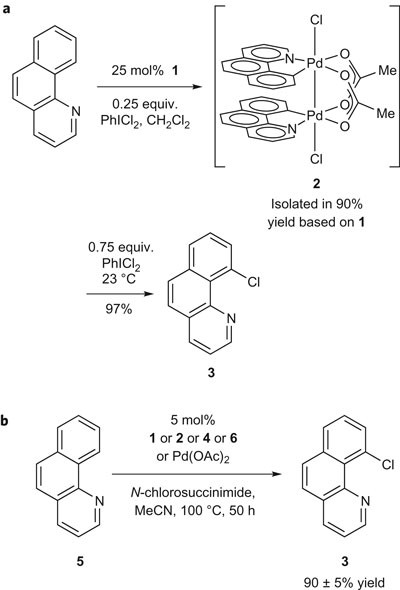
Bimetallic Pd(III) complexes in palladium-catalysed carbon–heteroatom bond formation | Nature Chemistry

Formation of palladium complexes of ligands 2a, 5 and 7. (A) Formation... | Download High-Quality Scientific Diagram
![PDF] Palladium-catalyzed C-C bond formation of arylhydrazines with olefins via carbon-nitrogen bond cleavage. | Semantic Scholar PDF] Palladium-catalyzed C-C bond formation of arylhydrazines with olefins via carbon-nitrogen bond cleavage. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/43c3b24a29c631438999caf7ba3027f907f307ae/17-Figure1-1.png)
PDF] Palladium-catalyzed C-C bond formation of arylhydrazines with olefins via carbon-nitrogen bond cleavage. | Semantic Scholar

The ubiquitous cross-coupling catalyst system 'Pd(OAc) 2 '/2PPh 3 forms a unique dinuclear Pd I complex: an important entry point into catalytically c ... - Chemical Science (RSC Publishing) DOI:10.1039/C9SC01847F
Synthesis of dipalladium(III) complexes with Pd-Pd bonds. Formation... | Download Scientific Diagram

Unexpected Formation of Hexasubstituted Arenes Through a Twofold Palladium-Mediated Ligand Arylation | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Palladium-catalyzed sequential carbon-carbon bond cleavage/formation producing arylated benzolactones. | Semantic Scholar

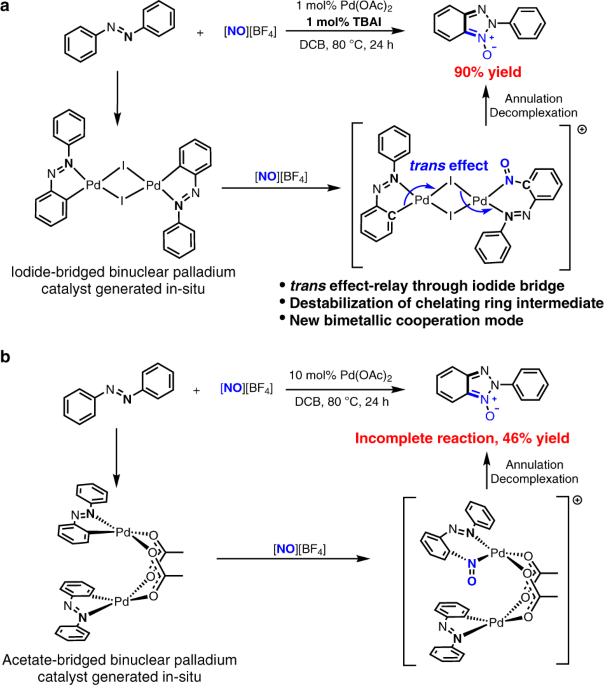
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex - ECHEMI
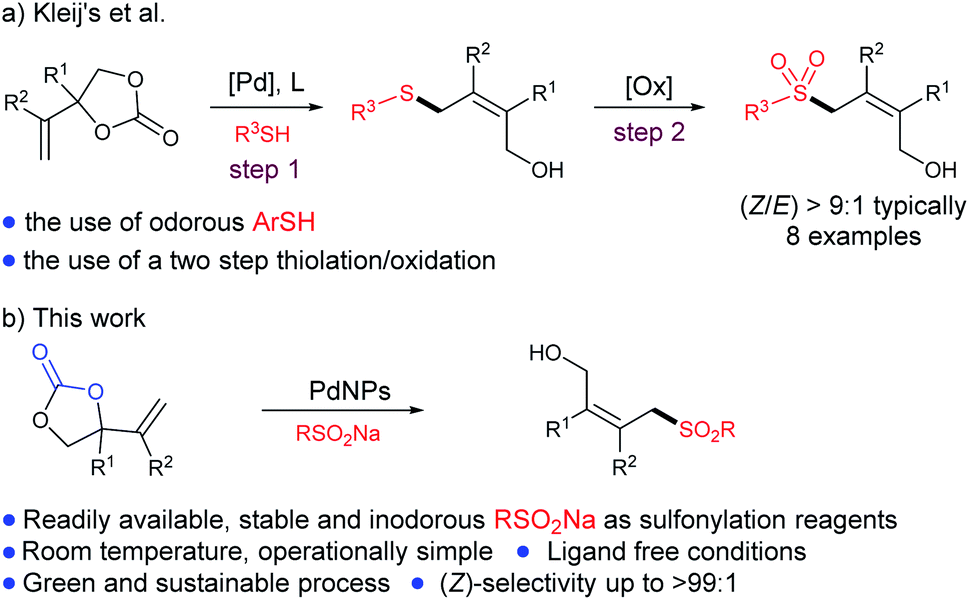
Palladium nanoparticles as efficient catalyst for C–S bond formation reactions - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05848C

Bimetallic Pd(III) complexes in palladium-catalysed carbon–heteroatom bond formation | Nature Chemistry

Scheme 1 A catalytic cycle for palladium(0)-mediated benzylphosphonate... | Download Scientific Diagram
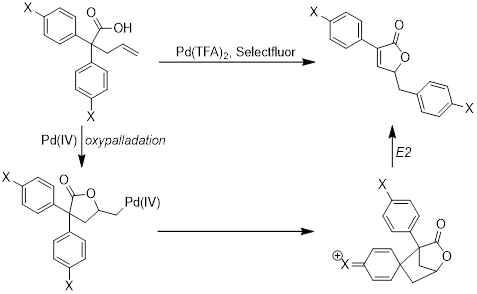
Report: Development of a Palladium-Catalyzed Oxidative Aryl Transfer Reaction (62nd Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

Homogeneous Palladium Catalyst Suppressing Pd Black Formation in Air Oxidation of Alcohols | Journal of the American Chemical Society

Enhancing stability by trapping palladium inside N-heterocyclic carbene-functionalized hypercrosslinked polymers for heterogeneous C-C bond formations | Nature Communications